 
To enable future reactors, researchers need theoretical modeling of the behavior of hydrogen and deuterium-tritium fuel, not just of deuterium fuel. Current fusion tokamaks use only deuterium because it is easier to acquire and handle than tritium. These isotopes will fuse together into helium atoms and release large amounts of energy. The Impactįuture fusion reactors will use a 50-50 mix of two hydrogen isotopes, deuterium and tritium. The research revealed an improved theoretical model that includes the tiny, but important, mass of the electron. A new analysis has helped unravel this mystery. That contradiction is the disconnect between theoretical predictions and experimental observations of how fusion energy confinement varies with the mass of hydrogen isotopes used to fuel the plasma. But plasma scientists have long been puzzled by a mysterious contradiction. These isotopes are the fuel for the fusion reaction. Used in making heavy water which is used for moderator in nuclear fission processes.Creating a fusion plasma requires deep understanding of the behavior of various isotopes of hydrogen. 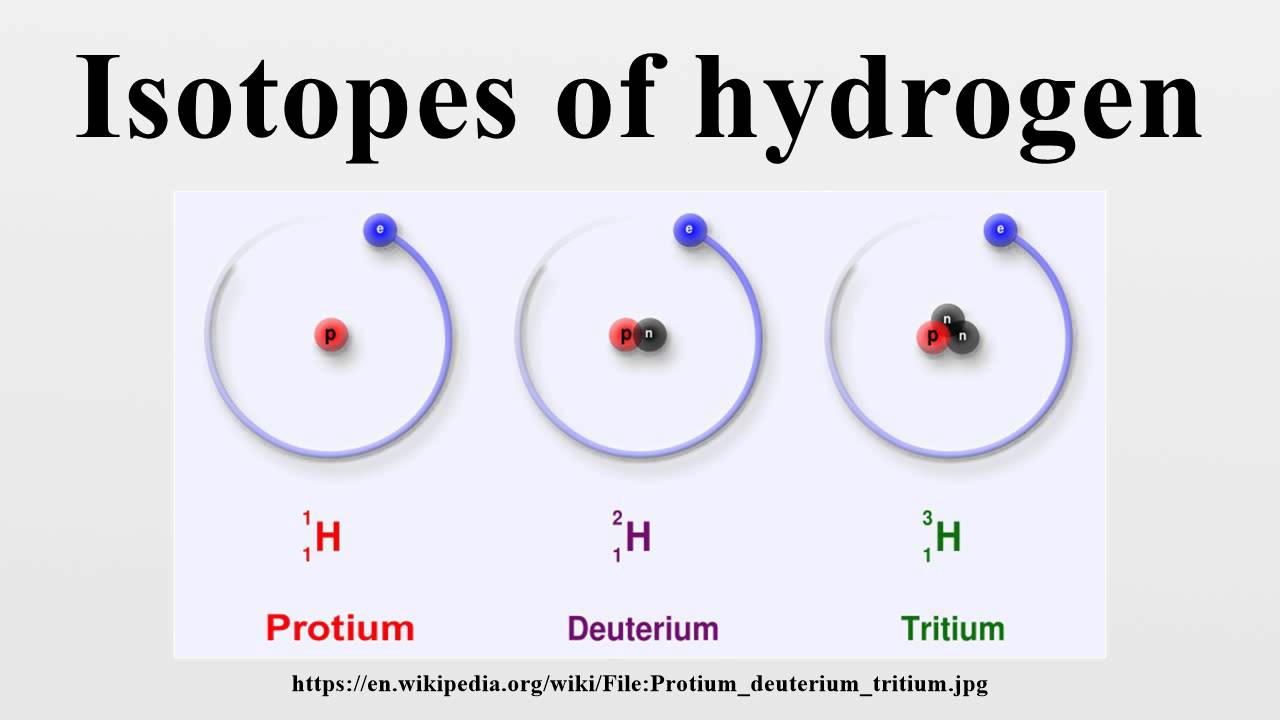
Uses ofĪs a projectile in nuclear reactions and as a reactor in nuclear fusionĪs a tracer element in the study of the processes of various reactions and Such reactions are called Exchange Reactions. In suitable conditions, it substitutes hydrogen from compounds of hydrogen by deuterium. It exhibits Summative reactions with unsaturated hydrocarbons. Burning:ĭuetro-Halides is formed by acting with halogens in appropriate positions.ģD 2 + N 2 –Fe+Mo → 2ND 3 (Deutero-ammonia) Sulfur:ĭ 2 + S –high temperature → D 2S (deuterium sulfide) Metals:ĢNa + D 2 – 350 0C → 2NaD (Sodium deuteride)Ĭa + D 2 – 400 0C → CaD 2 (Calcium deuteride) Summative reactions: The following are some of the key chemical reactions of It. The chemical reactions of hydrogen are about twice as fast as the chemical reactions of Deuterium. The speed of chemical reactions of Deuterium and hydrogen varies. Hence, Deuterium and hydrogen also have similar properties. The chemical properties of isotopes are similar. Its weight and density are two times the weight and density of hydrogen. It is a colorless, odorless and tasteless gas. This method was first used by Harold Urey in 1931. Light hydrogen (boiling point = 20.28 K) evaporates first and the Deuterium (boiling point = 23.59 K) remains in the distillation flask. In this method, liquid hydrogen is effectively distilled in a vacuum. By expanding ordinary hydrogen intoĪ porous pot, light hydrogen will expand rapidly. Light gases expand faster than heavy gases. Diffusion Method:Īccording to Graham’s law of diffusion, the diffusion speed of a gas is inversely the square root of its atomic mass. The following is the main method of separating duet from ordinary hydrogen. HydrogenĪbout 6,000 parts of ordinary hydrogen contain about 1 part deuterium. Reactions are used both for making small amounts of duet gas in the laboratoryĪnd for its industrial manufacture. And uses the electrode of nickel.ģ) Duet gas is also obtained when heavy water vapor flows over a blood-heated iron, zinc, or magnesium metal. For the decomposition of heavy water, small amounts of phosphorus pentaoxide or sodium carbonate are added to it. Deuterium gas is obtained.Ģ) The electrical decomposition of heavy water also gives duet gas. The main method of obtaining deuterium to heavy water is the following:ġ) Deuterium is obtained by the action of sodium from heavy water. The oxide (D2O) of Deuterium is also called heavy water. for the discovery of Deuterium, Harold Urey received the Nobel Prize in 1934. Like hydrogen, deuterium has two atoms in one Atom. Hence, about 1000 parts of ordinary hydrogen contain about 1 part of heavy hydrogen. Ordinary hydrogen contains 99.98% protium, 0.02% deuterium and an almost negligible amount of tritium. All the three sites of hydrogen, only the protium and deuterium are stable. Protium is also known as light hydrogen and deuterium as heavy hydrogen. 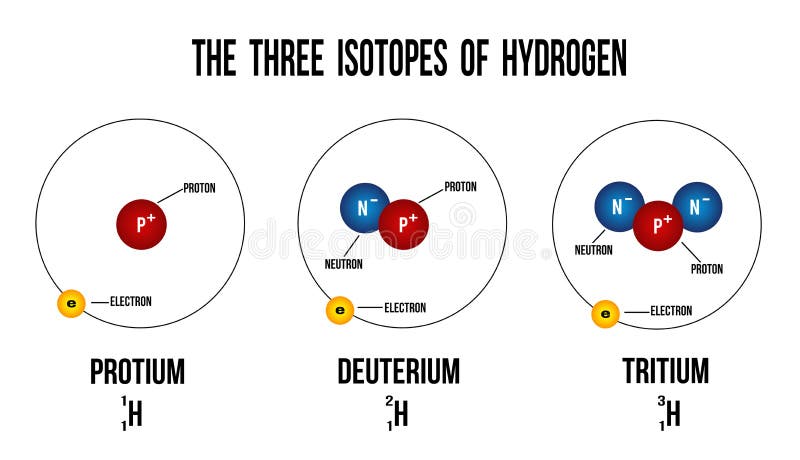
Each of them has one electron and one proton and the number of neutrons is 0, 1, 2 respectively. In chemical equations, they represent H, D, and T respectively. Their symbols are 1H 1, 1H 2, 1H 3 respectively. These institutes are called Hydrogen-1( protium), Hydrogen-2(deuterium) and Hydrogen-3( tritium). Their atomic number is 1 and their mass numbers are 1, 2, and 3 respectively. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
